The second column contains the numbers “1” and “1” as well as the merged cell.  
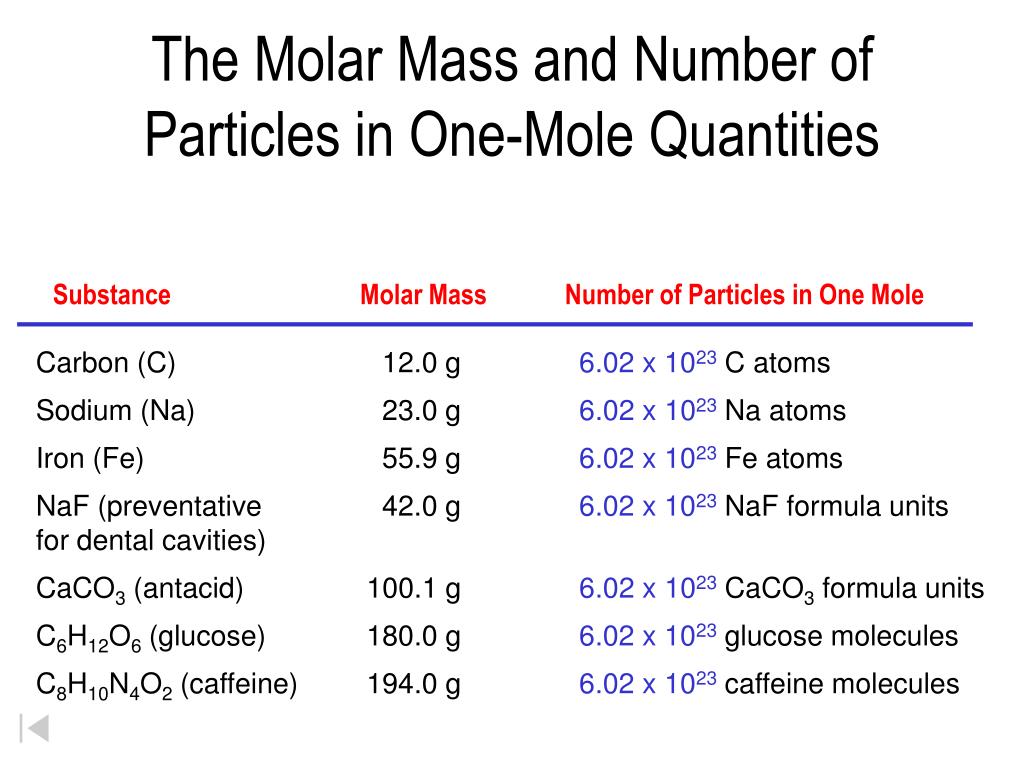
Pretend you have 400 g of NaCl in 1.5 L of H 2 O. The merged cell runs the length of the first five columns. If you dont know the moles but you have the mass, you can still figure out the molarity by using the mole ratio. The header row reads: “Element,” “Quantity,” a blank space, “Average atomic mass (a m u),” a blank space and “Subtotal (a m u).” The first column contains the symbols “N a”, “C l,” and a merged cell. The table is made up of six columns and four rows. We can compute the molar concentration by dividing the number of moles by the volume of the solution.\): Table salt, NaCl, contains an array of sodium and chloride ions combined in a 1:1 ratio. It, in turn, leads to the number of moles of the base. To find the number of moles, we shall divide it by the Avogadro number, 6×10 to get 0.00333 mol. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game An official website of the United States government. The balanced chemical equation always leads to a mole ratio between the acid and the base. For an element, the molar mass is the mass of 1 mol of atoms of that element for a covalent molecular compound, it is the mass of 1 mol of molecules of that compound for an ionic compound, it is the mass of 1 mol of formula units. Interactive periodic table with up-to-date element property data collected from authoritative sources. Add these together to get the molar mass of sodium hydroxide, which turns out to be 40 grams. For a molecule or compound, simply add up all the molar masses of the elements, taking subscripts into account. Rounding to a whole number, their molar masses are 23, 16 and 1 grams respectively. The molar mass of any element is on the periodic table. All mole calculations will determine the amount in moles of the solution, for which it is the molar concentration. From the periodic table, you find the atomic masses of sodium, oxygen and hydrogen to be 22.990, 15.999 and 1.008 amu respectively. Molarity is the number of moles of the solute dissolved per liter of the solution. Molar concentration is the most convenient method of expressing the concentration of a solute in the given solution. Source: en. Molar Concentration Formula Definition of Molar Concentration In this topic, we will discuss the formula for the Molar Concentration formula with examples. For KMnO4: Molar mass of K 39.1 g Molar mass of Mn 54.9 g Molar mass of O 16.0 g Molar mass of KMnO4 39.1 g + 54.9 g + (16.0 g x 4) Molar mass of KMnO4 158. From the periodic table the molar masses of the compounds will be extracted. To convert moles of a chemical to grams we need a conversion ratio which uses moles and grams: the molar mass of beryllium chloride. We will determine how many moles of a substance we have and then divide it by the volume of the solution. To convert grams to moles, the molecular weight of the solute is needed. Calculating the molar concentration of a solution is a comparatively straightforward process. Solution and its related computation are very important in science.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
